Microsurgical Tubal Reconstruction
Authors
INTRODUCTION
Tubal factor infertility, a significant cause of female infertility, results mostly from the consequences of pelvic inflammatory disease, endometriosis, and postoperative pelvic adhesions. Microsurgery remains an important operative technique for the management of tubal obstruction. It was introduced into gynecology primarily to improve the results of tuboplasty. In essence it involves magnification, the use of gentle tissue handling, fine sutures, meticulous methods for hemostasis, and copious irrigation. These principles are essential during laparoscopy or laparotomy. Operative laparoscopy using electrical, thermal, and laser energy, sharp and blunt dissection, ligation, and suturing techniques established the advantages of this approach for gynecologic procedures. Operating with a closed peritoneal cavity prevents drying of tissues, avoids the use of packs, provides a degree of magnification, and requires fewer days of postoperative hospitalization.
Nevertheless, many types of tuboplasty procedures remain in which a properly performed laparotomy is required, and it is the purpose of this chapter to emphasize this access to the pelvis. The choice among these procedures must be made by the patient after having been apprised of the expected outcomes (e.g., success rates, costs, potential complications) and the availability of experts and facilities. Patients who desire reversal of a previous sterilization in which adequate proximal and distal tubal segments remain are served best by tubal anastomosis at laparotomy, rather than a laparoscopic approach, or by assisted reproductive technologies. Novy1 augmented fimbriectomy reversal with concurrent gamete intrafallopian transfer and embryo cryopreservation during the same treatment cycle, followed by subsequent embryo thaw and transfer. He reported eight pregnancies in four patients in an original group of seven patients.
The selections of the most appropriate patient and the proper surgical procedure are essential for optimizing results. Operations should be discouraged in patients who have advanced tubal disease. Although in vitro fertilization has reduced the demand for reconstructive tubal operations, the repair of occluded tubes remains the first choice of most of these women.2
SELECTION OF PATIENTS
It is proper to do an infertility study that includes ovulation testing, semen evaluation, and a hysterosalpingogram (HSG) to be certain that only the tubal abnormality has caused the infertility. Contraindications to tuboplasty include a history or laboratory evidence of pelvic tuberculosis, advanced age of the woman, and recent pelvic inflammatory disease. A tuboplasty offers little chance of success in some patients with certain types of tubal abnormalities, such as those who have had a bilateral salpingectomy; those with bipolar, bilateral tubal obstruction; those with large hydrosalpinges (greater than 3 cm) and absent rugae; and those who have severe adnexal adhesions involving the ovaries.
The HSG should be fluoroscopically controlled under image intensification with water-soluble contrast material. Small increments of the contrast material are injected. The end points are either tubal fill and spill, or increasing abdominal pain in the absence of tubal filling. The drainage film is essential to ascertain the presence of spill or localization, and to evaluate rugae in the distally obstructed tube (Fig. 1). The association of salpingitis isthmica nodosa with a hydrosalpinx in the same oviduct represents a contraindication to further attempts at tubal reconstruction. Laparoscopy is employed for diagnostic confirmation and possible therapeutic procedures. Other endoscopic techniques that have become a part of the evaluation of the fallopian tube include salpingoscopy3 and falloposcopy,4 the latter utilizing a linear everting catheter.
TECHNIQUES
The performance of microsurgical procedures requires proper instruction and adequate practice to maintain a level of proficiency. The proper setting for initiating this undertaking is in the laboratory, not in the operating room. The basic mechanical task is to use fine sutures precisely and to approximate edges without distortion or tension. Training with the use of the binocular microscope affords the gynecologic surgeon the opportunity to adequately resect the abnormal tissue because of the instrument's ability to obtain a high degree of magnification, with least distortion and excellent illumination. The angled binocular oculars (X20) and objectives (200 mm) give a wide field of view. A foot-pedal or hand control can adjust the zoom and focusing. The assistant observes the procedure with opposite binoculars that offer the same choice of magnification. Loupes are designed to meet the requirements of the individual surgeon and can magnify to X6 at a distance of 380 mm.
Microsurgical instruments such as forceps, needle holders, and scissors are lightweight and approximately 13 to 15 mm in length. Instruments are sterilized and packaged in a special container and should be inspected frequently for imperfections.
Fine sutures (7-0 to 10-0) must be tied gently so that they do not break, and with at least three knots so that they do not loosen. Needles should be tapered, have a curvature of approximately 135°, and a diameter of 50 to 150 μm.
The electrosurgical generator is set at the lowest current to provide adequate hemostasis and cutting, as required. Microelectrodes are available for dissection and removal of adhesions, scalpel-like cutting, and pinpoint coagulation with minimal charring. The handles should have fingertip controls.
The surgeon should be familiar with a range of infertility disorders and have experience with microsurgical techniques. During the vaginal preparation, an intrauterine cannula device is inserted so that intraoperative chromopertubation can be done as needed. The patient should be in a moderate Trendelenburg position with a comfortable lateral tilt toward the surgeon. A transverse suprapubic incision will afford maximal exposure to the pelvic structures. The surgeon should sit opposite the tube to be treated whenever there are significant periadnexal adhesions to the posterior leaf of the broad ligament or the cul-de-sac associated with distal tubal obstruction. This position enables the surgeon to see the involved area more clearly. Sharp, careful dissection will lessen the chance of inadvertent injury to vital structures and reduce unnecessary peritoneal and serosal trauma. The uterus should be elevated to the level of the abdominal incision and the adnexa stabilized on wet laparotomy pads and Kerlex packing. Procedures should not be prolonged because cells and tissues can be damaged irreversibly because of desiccation. The floor-mounted microscope and the operating table should not interfere with the surgeon's access to the pelvic structures and ability to manipulate the microsurgical instruments.
CLASSIFICATION OF TUBOPLASTIC PROCEDURES
- Lysis of periadnexal adhesions (classify according to the adnexa with the least abnormality)
- Minimal: 1 cm of the tube and ovary involved (less than one third)
- Moderate: Partially surrounding the tube or ovary
- Severe: Encapsulating peritubal and/or periovarian adhesions (more than two thirds)
- Minimal: 1 cm of the tube and ovary involved (less than one third)
- Lysis of extragenital adhesions
- Minimal
- Moderate
- Severe
- Minimal
- Tubo-uterine implantation
- Isthmic: Implantation of the isthmic segment
- Ampullary: Implantation of the ampullary segment
- Combination: Different types of implantations on opposite sides
- Isthmic: Implantation of the isthmic segment
- Tubotubal anastomosis
- Interstitial (intramural)-isthmic
- Isthmic-isthmic
- Isthmic-ampullary
- Ampullary-ampullary
- Ampullary-infundibular (fimbrial)
- Combination: Different types of anastomoses on right and left tubes
- Interstitial (intramural)-isthmic
- Salpingostomy (salpingoneostomy): Surgical creation of a new tubal ostium
- Terminal
- Ampullary
- Isthmic
- Combination: Different types of salpingostomy on right and left tubes
- Terminal
- Fimbrioplasty
- Deagglutination and dilation
- Serosal incision for completely occluded tube
- Combination: Different types of fimbrioplasty procedures on right and left tubes
- Deagglutination and dilation
- Other reconstructive tubal operations (type to be specified)
- Combinations of different types of operations
- Bipolar: For occlusion of proximal and distal ends (types to be specified)
- Bilateral: Different operations on the right and left tubes
- Bipolar: For occlusion of proximal and distal ends (types to be specified)
This classification is based on the type of tubal repair, regardless of the cause of the occlusion.
DISTAL TUBAL OBSTRUCTION
The surgical management of distal tubal obstruction can be done by laparotomy or laparoscopy with almost similar results. The choice of access depends on several factors, including the skill of the surgeon with each approach and the degree of tubal abnormality. The procedure that gives the best prognosis and is easiest to perform is lysis of adnexal adhesions. This operation can be done laparoscopically, most often using a three-puncture technique. The areas being dissected must be kept in the center of the field, as seen on the monitor, and the adhesions are held under tension. Usually these adhesions can be cut with laparoscopic scissors, which should be sharp to avoid tearing. An electrocoagulating needle electrode is needed infrequently. With a laparotomy, the surgeon should stand opposite the adherent adnexum to reduce injury to the posterior leaf of the broad ligament and the ureter. Mobilization of the tube and ovary is done under direct vision by sharp dissection. Patience and perseverance will be rewarded by this approach because there will be minimal repair required of the separated structures. Peritoneal defects are not sutured. The cut adhesions are removed and the areas irrigated constantly to avoid desiccation.
With a terminal salpingoneostomy, once the fallopian tube has been freed from all adhesive bands either laparoscopically or at laparotomy, a white dimple-scarred area is identified at the end of the fallopian tube. Incisions are made into these lines with either an electrosurgical needle electrode or a carbon dioxide laser.5 Tubal flaps created by these incisions can be sewn back to the tubal serosa with a few free sutures. Alternatively, the area behind these flaps can be desiccated, causing the edges to fold back and evert the endosalpinx. This maneuver often reduces the need for suturing. Then the endosalpinx can be evaluated as to its color, consistency, and presence of mucosal adhesions and rugae. These findings influence the prognosis for fertility. In their study of the results from salpingoneostomies, Vasquez and associates6 found that the thin-walled hydrosalpinges without adhesions showed a 53% pregnancy rate. In the thin-walled hydrosalpinges with mucosal adhesions, the intrauterine pregnancy rate diminished to 23% and the ratio of tubal to intrauterine pregnancy was increased. In 1988, the American Fertility Society7 proposed a scoring system based on the extent and type of adhesions. For the evaluation of distal tubal occlusion, the thickness and rigidity of the tubal wall, its ampullary diameter, and percentage of mucosal folds preserved at the site of the neostomy are described. Intraoperative salpingoscopy was recommended to see the entire length of the ampullary lumen, but was not included in the scoring system. Marana and co-workers8 found that only the status of the mucosa correlated significantly with the subsequent pregnancy outcome. Salpingoscopy provided more accurate prognostic information than gross assessment of the mucosa at the site of the neostomy. During microsurgery or laparoscopy, when mucosal folds appeared more than 75% preserved at neostomy, the ampullary mucosa appeared normal at salpingoscopy in only 30% (9/30) of the patients. When the mucosal folds seemed 35% to 75% preserved, salpingoscopic findings were normal in 32% (7/22) of the patients.
Laparoscopic salpingoneostomies were done on 87 infertile women using either scissors and thermocoagulation for eversion or the carbon dioxide laser.9 Cumulative pregnancy rates were evaluated by life-table analysis according to the tubal classification and the mucosal status. The 18-month estimated cumulative pregnancy rate with normal delivery was 28.7%. Pregnancy rates were significantly higher in patients classified as grades I and II compared to grades III and IV (severely damaged tubes).10
Midampullary salpingoneostomy is done in few patients. The width of the distally occluded end should be at least 1 cm if any success is to be realized. When the cause of the occlusion is the result of a partial salpingectomy for an ectopic pregnancy or an attempt to reverse a previous sterilization, the prognosis is better than if the tube had been occluded because of pelvic inflammatory disease. Isthmic salpingoneostomy, the creation of a new opening in the isthmic tubal segment, in a tube 5 cm or less in length, should not be done because the chance of a postoperative pregnancy is nil. As in all tubal procedures, the operative sites are irrigated with heparinized lactated Ringer's solution to avoid desiccation and to reveal small sites of bleeding that may require coagulation. Artificial devices are no longer advocated to maintain the neostomy.
Fimbrioplasty is designed to restore occult fimbriae to their normal position. Sometimes the fimbriae are hidden completely so that the tube has the appearance of a hydrosalpinx. Careful palpation of such a tube will reveal a terminal nodularity caused by bunched, trapped fimbriae. Lysis of the serosal covering often will allow the trapped fimbriae to escape from their entrapment. Minimal bleeding is encountered in this operation. Another variation of fimbrioplasty is deagglutination or dilation of a phimotic infundibular ostium. The results for laparoscopic fimbrioplasty are similar to those for laparotomy. Many reports fail to distinguish between fimbrioplasty and salpingoneostomy.
PROXIMAL TUBAL OBSTRUCTION
The causes of proximal tubal obstruction include salpingitis isthmica nodosa, obliterative fibrosis, endometriosis, and follicular salpingitis. Endotubal synechiae, mucus, debris, and cornual spasm can simulate organic proximal tubal obstruction.
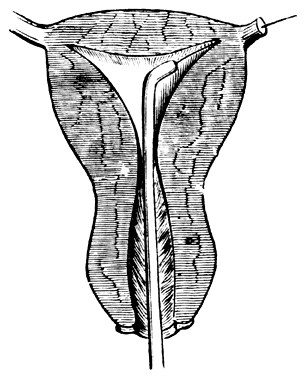
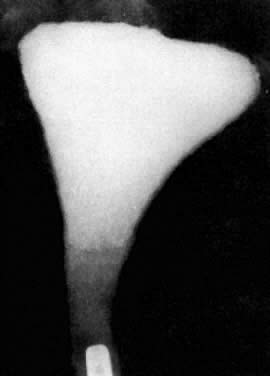
Attempts to overcome proximal tubal obstruction date from the middle of the 19th century. Metal probes were inserted transcervically until it was felt that the intramural tubal segment was reached and then presumably these probes were advanced until the “obstruction” was overcome (Fig. 2). Until the use of the HSG became a part of the infertility work-up, the diagnosis of proximal tubal obstruction could only be suspected. In this condition the intramural (interstitial) segment that connects the cornua to the isthmus usually is patent. On the HSG, the contrast material should show pointed rather than rounded ends (Fig. 3); failure of the tubes to opacify with contrast material is presumptive evidence of proximal obstruction, but tubal spasm, synechiae, and “debris” can simulate organic occlusion. In analyzing the HSG, it is important to remember that the length of the intramural segment is about the width of the thumb. A thumb placed at the cornua on the radiograph will cover the width of the myometrium (Fig. 4).11
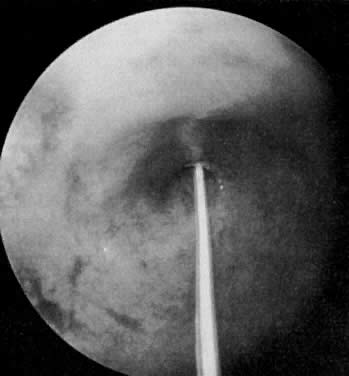
Laparoscopy with chromopertubation is required to prove the diagnosis. The next procedure to be undertaken at this juncture is a transcervical tuboplasty. Most reports have documented the feasibility of transcervical tubal cannulation as a diagnostic and therapeutic procedure that in some cases could replace an abdominal tuboplasty.12 This procedure does not require extensive preoperative preparation or laboratory evaluation. It can be done under analgesia, intravenous sedation, or a paracervical block. Fluoroscopic, sonographic, or endoscopic control (Fig. 5) can be used. Grow and colleagues13 reviewed the published series on transcervical tubal cannulation until 1993, describing its success in establishing patency and pregnancy rates. They also noted its use for deposition of gametes and embryos and for direct observation of the endosalpinx.
Macrosurgery has been replaced by microsurgical techniques, the latter requiring a laparotomy to achieve the best results. Once it is clear that the proximal obstruction cannot be overcome by transcervical cannulation and that the distal tubal segment is grossly normal, the surgeon can elect to do a tubal anastomosis by resecting the occluded portion of the tube. Proximal obstructions occur mostly either at the junction of the interstitial and isthmic segments or in the tubal isthmus. Tubotubal anastomoses are made between equal or unequal tubal segments, and each type requires special consideration.14 The intramural segment has the smallest lumen and the ampullary the widest, with a thin myosalpinx. Only after patency has been established in the remaining resected segments and their ends examined microscopically to ensure that they are normal can the anastomosis begin. To reduce tension on the anastomosis, a suture in the myosalpinx is used to approximate the tubal segments. When the anastomosis involves the intramural segment, four mucosal folds should be identified and free-flowing indigo carmine is expected. Injection of a dilute solution of vasopressin into the cornual region with a fine-gauge needle helps to reduce the oozing from this region. The posterior 6 o'clock suture is placed initially in the distal segment from the serosal side toward the edge of the lumen. Then it is sutured to the proximal segment so that the knot is external to the lumen. Usually a two-layered anastomosis is done and tubal patency tested. Some leakage may be seen at the site of the anastomosis, although a true test of patency is observation of dye at the fimbriated end. These general principles apply to most types of anastomoses. There is evidence to suggest that tubal length is related directly to a successful outcome, and there is an inverse relationship between the final length and the rapidity of conception. Although laparoscopists have done tubotubal anastomoses, the results are not as good as those reported from microsurgical procedures. A study of microsurgical anastomosis on the rat uterine horn, in which two, four, and eight sutures were used, compared the results after 60 days. Since no differences were noted in the serosal, muscularis, or endosalpingeal layers in any of the groups, it would appear that two sutures were adequate for tissue repair.15
Tubo-uterine Implantation
If fibrosis replaces the entire interstitial tubal segment, then implantation is required. This condition is rare, so implantations are done infrequently. The advent of the surgical microscope has made tubocornual anastomosis the preferred procedure for the management of proximal tubal obstruction involving the interstitial isthmic segments, such that a tubal implantation procedure has become of historic interest. The advantages of tubocornual anastomosis over tubal implantation are as follows:
There is less bleeding.
Less uterine and tubal tissue is removed.
There is a reduced chance of uterine rupture should pregnancy result.
Cesarean delivery is not needed because the uterine wall is not weakened.
Implantation remains a possibility should tubocornual anastomosis fail.
Reversal of Tubal Sterilization
The fundamental operation for reversal of a tubal sterilization is a microsurgical anastomosis. Success of the procedure depends on the extent of tubal destruction and the length of the repaired tubal segments. If the reconstructed tube has a length of at least 4 cm with an ampullary length of 1 cm, live birth rates of 60% to 80% can be achieved. The tubal pregnancy rates vary, but average 2% to 5%.
Kroener sterilization (fimbriectomy) has been considered irreversible, but if at least 50% of the ampulla has been preserved an ampullary salpingoneostomy will yield a significant live birth rate.
Experimental studies to evaluate the effectiveness of several types of anastomoses including the use of fibrin adhesive,16,17,18 tissue-adhesive cyanoacrylate,19 and laser welding20 have been used on animals to evaluate the efficacy of the procedures. The time required for the procedures, the degree of adhesion formation, and the patency of the anastomosis were evaluated, and the quality of the scars were examined grossly and histologically. Each procedure appeared to have severe limitations, either because of foreign body granulomatous reactions or because it was unsuitable for certain types of anastomoses, notably the tubocornual procedures. Laparoscopic tubal anastomosis was done on five previously sterilized patients, and patency was documented in 5 of the 10 tubes. The crude pregnancy rate was 50%.21
The most frequent reason to do a tubal anastomosis is to reverse a previous sterilization. Careful screening with the inclusion of a preliminary laparoscopy is essential if the type of sterilization is not known, if there is no pathology report of an extirpated specimen, or if electrosurgery was used to sterilize the patient. Most patients become pregnant within 2 years after the reversal. If pregnancy does not occur within 6 months of the operation, an HSG is suggested to detect the patency of the anastomosis. There is evidence to show a direct relationship between the length of the repaired tube and the time required for a postoperative conception. Pregnancy rates seem to be better in patients who have undergone removal of mechanical devices used for sterilization (e.g., clip or falope ring) than in patients who have undergone reversal of electrosurgical sterilization.
ADJUNCTIVE THERAPY
One major factor causing failure to conceive postoperatively is the formation of pelvic adhesions. Several procedures have been employed to reduce this complication. The use of magnification, minimal and gentle tissue handling, perfect hemostasis, constant irrigation, and fine nonreactive sutures all help reduce the incidence of postoperative adhesions. Artificial devices, early and late second-look laparoscopy, hydropertubation, prophylactic antibiotics, and various anti-inflammatory agents have not produced significant improvement in results. Ideally, a physical barrier for the prevention of adhesions should be nonreactive, persist during reepithelialization of the reconstructed tube, resist bacterial growth, and undergo absorption appropriately. Oxidized, regenerated cellulose barriers appear to satisfy these criteria. Its efficacy has been evaluated during salpingolysis and fimbrioplasty to prevent the formation or reformation of postoperative adhesions.22 Adhesiolysis was done in 66 infertility patients who had peritubal adhesions.23 This barrier (Interceed, Johnson & Johnson, Arlington, TX) was applied to one side only, and a follow-up laparoscopy after 4 to 10 weeks showed twice as many tubes free of adhesions with the use of the barrier compared to tubes that had microsurgery alone.
COMPLICATIONS
Most patients who undergo tubal reconstructive surgical procedures can be discharged within 48 hours after surgery. Morbidity is uncommon, and prophylactic antibiotics are used infrequently because there is no evidence that they improve tubal patency or pregnancy rates. Instances of femoral neuropathy have been reported,24,25 but the major postoperative complications are a tubal pregnancy and reocclusion of the previously opened tube. A patient who becomes pregnant postoperatively must be watched carefully to exclude the possibility of a tubal pregnancy. A good precaution is to assume that there is a tubal pregnancy until an intrauterine gestation is proved. During the preoperative counseling, the patient should be made aware of the increased possibility of an extrauterine pregnancy.
RESULTS
The HSG was used to assess tubal patency in the postoperative evaluation of the infertile patient. Letterie and associates26 correlated the HSG and laparoscopic findings in 25 patients who had tubal surgery (microsurgical tubal anastomoses) and distal salpingoneostomies.14 HSG was more reliable in assessing tubal patency (sensitivity and specificity of 96% and 61%, respectively) than in detecting pelvic adhesive disease (sensitivity and specificity of 12% and 75%, respectively), regardless of the surgical procedure. HSG was associated with a high false-negative rate (60%), primarily because of its inability to detect pelvic adhesive disease. Complete agreement between HSG and laparoscopy was seen in only 15%. These data suggest that the HSG is a sensitive test to detect the presence of tubal patency, but was not sufficiently sensitive or specific to detect pelvic adhesive disease postoperatively. These limitations should be noted in the interpretation of the HSG in an infertile patient who has had tubal surgery.
Transcervical canalization was performed in 12 patients who developed occlusion of the proximal fallopian tube after either a failed reversal of sterilization7 or a tuboplasty to treat occluded fallopian tubes.12,27 The standard technique was attempted. Four of the seven patients with a failed reversal had fistulas, and one also had a stricture. The remaining 3 patients and all 12 patients treated after failed tuboplasty and tubal implantation had strictures at the site of the implantation or anastomosis. Transcervical recanalization failed in all patients with fistulas but succeeded in 13 of 15 tubes with stenoses. Three patients became pregnant 1 to 16 months after recanalization and two patients after in vitro fertilization and embryo transfer. Reocclusion occurred in 2 of 10 patients reexamined 6 to 36 months after recanalization.
SUMMARY
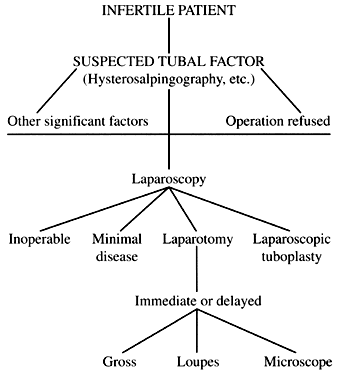
Tuboplasty remains an excellent option for selected patients who have a tubal factor as a cause for their infertility (Fig. 6). The properly counseled patient will be able to choose from a variety of options that include laparoscopy, laparotomy, assisted reproductive technologies, and in vitro fertilization. None of these procedures are mutually exclusive. The objective for the gynecologist is to direct the patient to the technique that will offer the greatest chance of achieving an intrauterine pregnancy.
REFERENCES
Novy MJ: Concurrent tuboplasty and assisted reproduction. Fertil Steril 62: 242, 1994 |
|
Holst N, Maltau JM, Forsdahl F et al: Handling of tubal infertility after introduction of in vitro fertilization changes and consequences. Fertil Steril 55: 140, 1991 |
|
Dunphy B, Pattinson HA: Office falloposcopy: A tertiary level assessment for planning the management of the infertile women. Aust NZ J Obstet Gynecol 34: 189, 1994 |
|
Kerin JF, Williams DB, San Roman GA et al: Falloposcopic classification and treatment of fallopian tube disease. Fertil Steril 57: 731, 1992 |
|
Nezhat CR, Nezhat FR, Luciano AA et al: Operative Gynecologic Laparoscopy: Principles and Techniques, pp 188 - 196. New York, McGraw-Hill, 1995 |
|
Vasquez G, Boeckx W, Brosens I: No correlation between peritubal and mucosal adhesions in hydrosalpinges. Fertil Steril 64: 1032, 1995 |
|
The American Fertility Society: The American Fertility Society classifications of adnexal adhesions, distal tubal occlusion, tubal occlusion secondary to tubal ligation, tubal pregnancies, müllerian anomalies and intrauterine adhesions. Fertil Steril 49:944, 1988 |
|
Marana R, Rizzi M, Mudii E et al: Correlation between the American Fertility Society classifications of adnexal adhesions and distal tubal occlusion, salpingoscopy, and reproductive outcome in tubal surgery. Fertil Steril 64: 924, 1995 |
|
Canis M, Mage G, Poully JL et al: Laparoscopic distal tuboplasty: Report of 87 cases and a 4-year experience. Fertil Steril 56: 616, 1991 |
|
Dubuisson JB, Chapron C, Morice P et al: Laparoscopic salpingostomy: Fertility results according to the tubal mucosal appearance. Human Reprod 9: 334, 1994 |
|
Hunt RB, Siegler AM: Hysterosalpingography: Techniques and Interpretation, p 105. Chicago, Year Book, 1990 |
|
Risquez F, Confino E: Transcervical tubal cannulation, past, present, and future. Fertil Steril 60: 211, 1993 |
|
Grow DR, Coddington CC, Flood JT: Proximal tubal occlusion by hysterosalpingogram: A role for falloposcopy. Fertil Steril 60: 170, 1993 |
|
Gomel V, Rowe TC: Microsurgical tubal reconstruction and reversal of sterilization. In Wallach EE, Zacur HA (eds): Reproductive Medicine and Surgery. St. Louis, CV Mosby, 1995 |
|
Caballero-Gomez JM, Ortega-Moreno J: Study of two simplified microsurgical techniques for uterine horn anastomosis in rat. Arch Gynecol Obstet 252: 191, 1993 |
|
Gauwerky JF, Klose RP, Vierneisel P et al: Fibrin glue for reanastomosis of the fallopian tube in the rabbit: Adhesions and fertility. Human Reprod 7: 1274, 1992 |
|
Weis-Fough US, Pedersen H, Schroeder E et al: Histomorphological evaluation of wound healing of rabbit oviduct after microsurgical reanastomosis with the use of autologous fibrin adhesive, human fibrin adhesive, or polyglycolic acid suture. Eur Surg Res 25: 278, 1993 |
|
Tulandi T: Effects of fibrin sealant on tubal anastomosis and adhesion formation. Fertil Steril 56: 136, 1991 |
|
Haj N, Haj M, Shasha SM et al: Tubal anastomosis in the rat using the tissue adhesive cyanoacrylate (Histoacryl). Gynecol Obstet Invest 38: 54, 1994 |
|
Kao LW, Giles HR: Comparison of laser-assisted anastomosis, laser welding, and microsurgical anastomosis of the rabbit uterine tubes. Obstet Gynecol 81: 122, 1993 |
|
Katz E, Donesky BW: Laparoscopic tubal anastomosis: A pilot study. J Reprod Med 39: 497, 1994 |
|
Azziz R: Microsurgery alone or with Interceed absorbable adhesion barrier for pelvic sidewall adhesion reformation. Surg Gynecol Obstet 177: 135, 1993 |
|
Nordic Adhesion Prevention Study Group: The efficacy of Interceed (TC7) for reformation of postoperative adhesions on ovaries, fallopian tubes, and fimbriae in microsurgical operations for fertility. Fertil Steril 63:709, 1995 |
|
Vanrell JA, Balasch J: Bilateral femoral neuropathy after microsurgical reversal of tubal sterilization: Case report and analysis of contributing factors. Human Reprod 2: 345, 1987 |
|
Hassan AA, Reiff RH, Fayez JA: Femoral neuropathy following microsurgical tuboplasty. Fertil Steril 245: 889, 1986 |
|
Letterie GS, Haggerty MF, Fellows DF: Sensitivity of hysterosalpingography after tubal surgery. Arch Gynecol Obstet 251: 175, 1992 |
|
Lang EK, Dunaway HH: Transcervical recanalization of strictures in the postoperative fallopian tube. Radiology 191: 507, 1994 |